NEW YORK (1010 WINS) -- The Food and Drug Administration on Tuesday issued an emergency use authorization for the first over-the-counter fully at-home diagnostic COVID-19 test.
Using a nasal swab, results will be available in 20 minutes.
The Ellume COVID-19 Home Test will be available for non-prescription home use for the detection of active COVID-19 in individuals with or without symptoms, and in adults and children aged 2 years and above, Ellume said in a press release Tuesday, announcing the FDA's approval.
Ellume is an Australian-based digital diagnostics company.
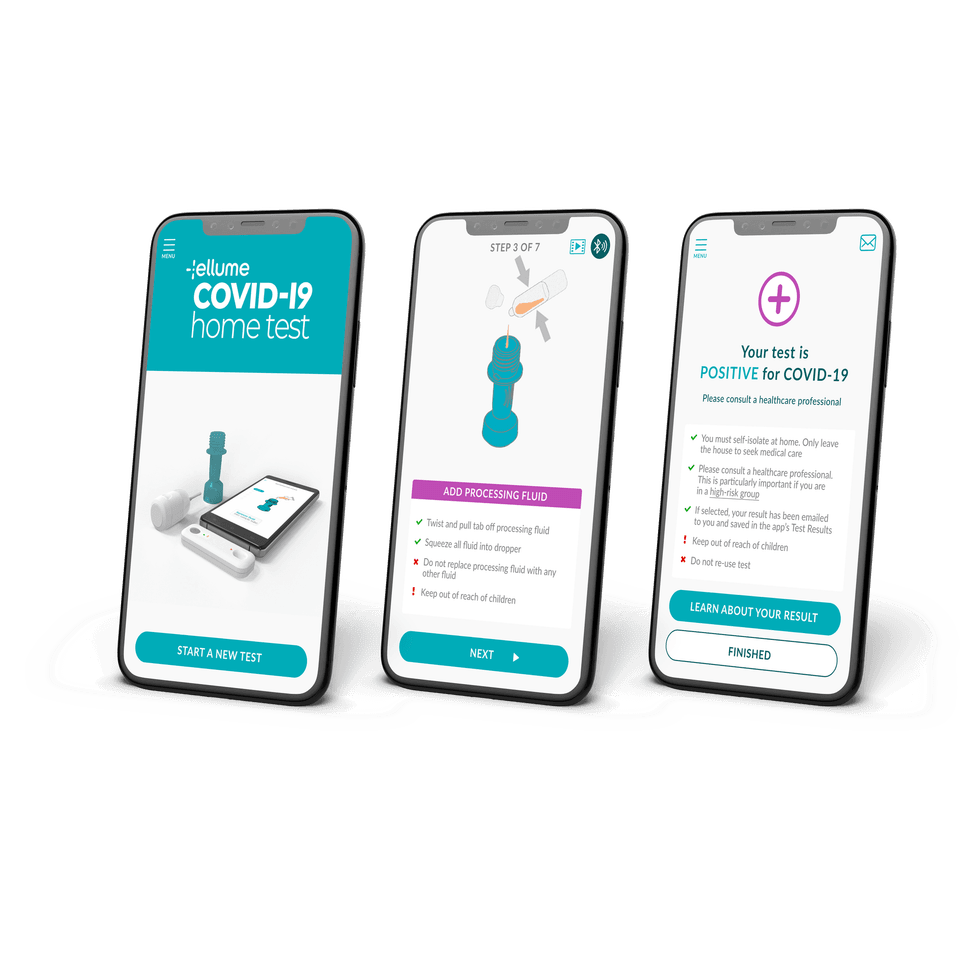
According to the FDA, the Ellume COVID-19 Home Test correctly identified 96 percent of positive samples and 100 percent of negative samples in individuals with symptoms. In people without symptoms, the test correctly identified 91% of positive samples and 96 percent of negative samples.
FDA Commissioner Dr. Stephen Hahn said in a statement, "Today's authorization is a major milestone in diagnostic testing for COVID-19. By authorizing a test for over- the-counter use, the FDA allows it to be sold in places like drug stores, where a patient can buy it, swab their nose, run the test and find out their results in as little as 20 minutes. As we continue to authorize additional tests for home use, we are helping expand Americans' access to testing, reducing the burden on laboratories and test supplies, and giving Americans more testing options from the comfort and safety of their own homes."








