Perrigo Company has issued a voluntary recall of 16,500 cans of store brand Premium Infant Formula with Iron Milk-Based Powder due to higher than permitted levels of Vitamin D.
The recalled product was shipped to CVS stores in 12 states: SC, TN, TX, FL, CA, VA, IN, NJ, MI, PA, RI, MO.

H-E-B Grocery in TX also received the formula.
The CVS Health brand Infant Premium formula shipped to stores in February of this year, 2024.

The bottom of the can has a use-by date stamp of November 11, 2025.
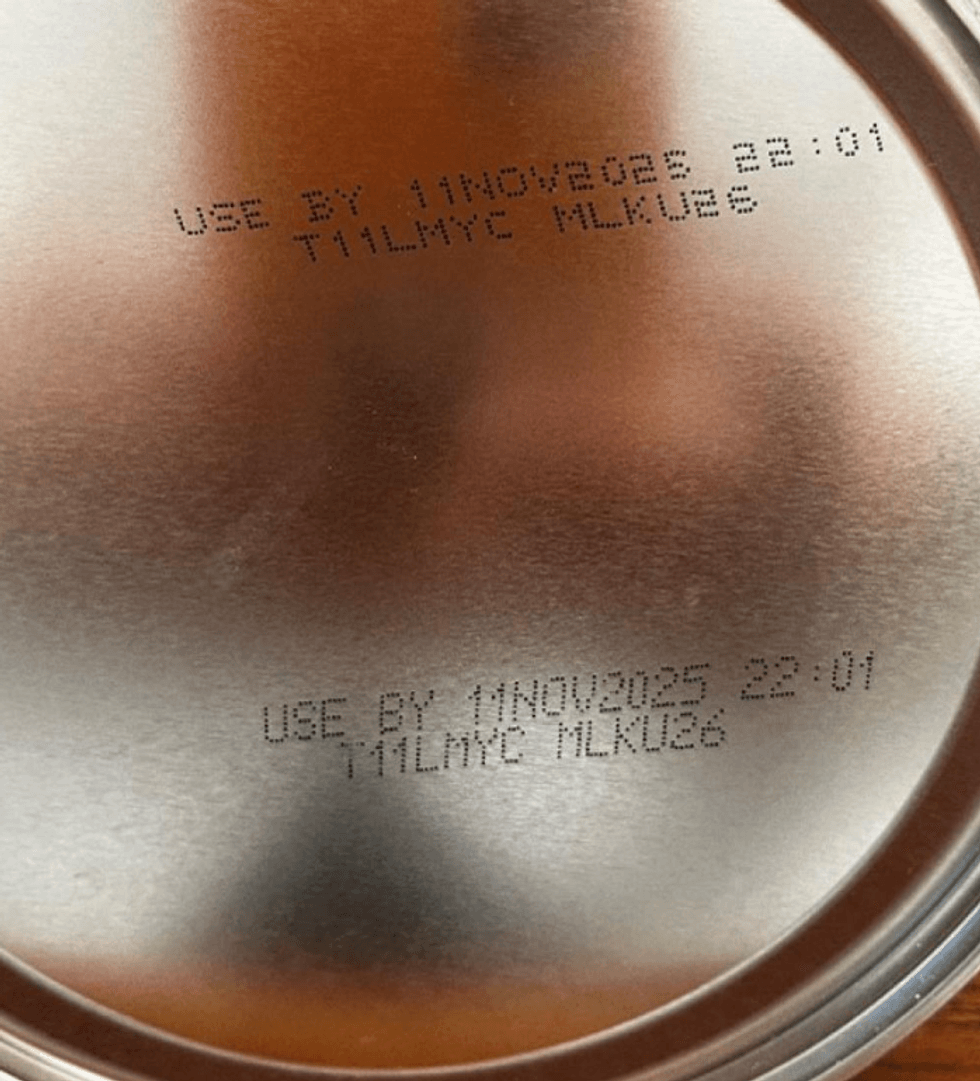
According to the U.S. Food and Drug Administration website, there have been no reports of adverse events tied to the recall, to date.
Any questions or concerns regarding the recall or adverse events associated with these lot codes can be communicated to Perrigo Consumer Affairs at 1-800-538-9543, M-F from 8:00am-5:00pm EST.







