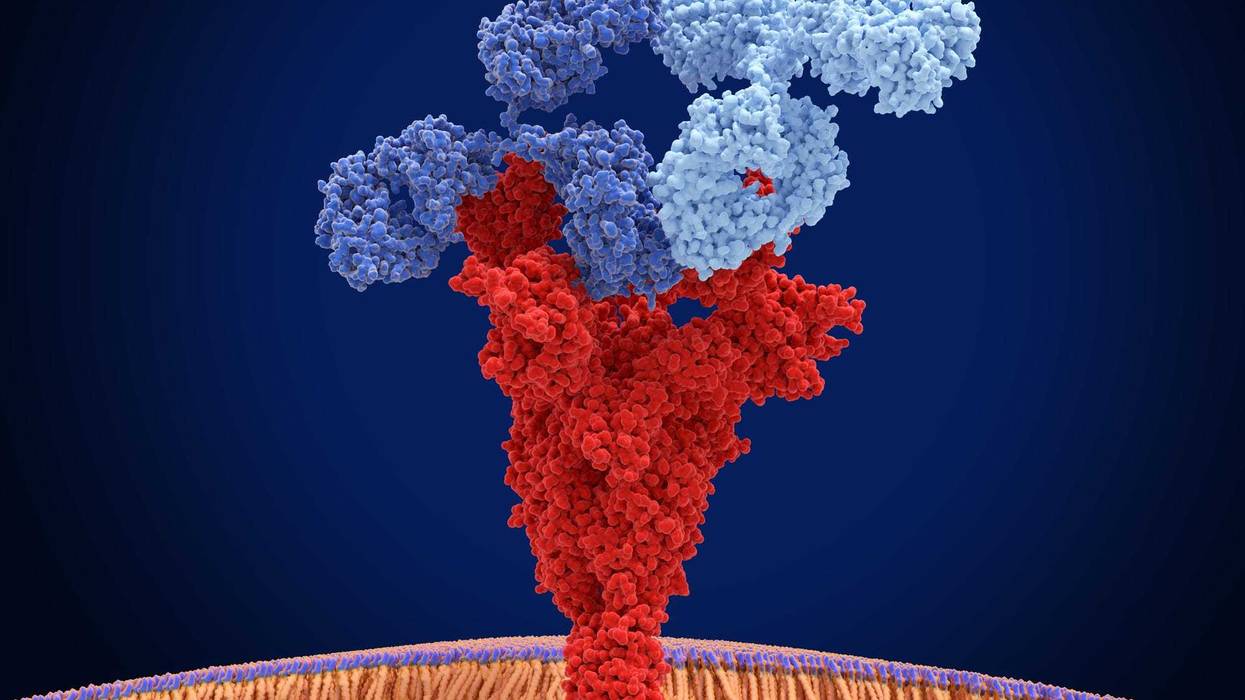
After receiving infusions of lab-made antibodies, some COVID-19 patients had a lower risk of hospitalization, according to a Mayo Clinic study released Monday.
The study was published in The Lancet's EClinicalMedicine journal.
Close to 1,400 Mayo Clinic patients were enrolled in the study, including 696 who received the treatment and an equal cohort who did not, said the Mayo Clinic. Observational research was conducted between December 2020 and early April, and patients were evaluated at 14, 21 and 28 days after treatment.
“At each point, the numbers for hospitalization were significantly lower in the treated group,” according to the clinic.
By the end of 28 days, 1.6 percent of those treated was hospitalized and 4.8 percent of those who had not been treated were hospitalized, around a 60 to 70 percent reduction in hospitalization for treated patients. ICU admission and mortality were low for those who received the antibodies.
“Our conclusion overall at this point is that monoclonal antibodies are an important option in treatment to reduce the impact of COVID-19 in high-risk patients,” says Dr. Raymund Razonable, a Mayo Clinic infectious disease specialist.
Monoclonal antibody treatments can help patients with mild to moderate symptoms who are at high risk of serious COVID-19 illness due to other health conditions, explained the Star Tribune. High risk patients include people age 65 and over as well as people with a high body mass index, chronic kidney disease or diabetes.
Mayo Clinic was the first health system in Minnesota last year to offer certain COVID-19 patients the antibody infusions, the Star Tribune added. Unlike vaccines, which aim to prevent are decrease the likelihood of infection, monoclonal antibodies are administered shortly after patients contract the virus. The treatment is designed to block virus from attaching to and entering human cells.
Late last year the treatment made headlines when then President Donald Trump received it after he was diagnosed with COVID-19. Though the federal government has been providing free access to drugs used in the treatment since November and December, initial demand from patients was underwhelming, said the Star Tribune.
Patients in the Mayo Clinic received a combination of casirivimab and imdevimab, antibody treatments that the Food and Drug Administration has cleared for emergency use, said the outlet.
As COVID-19 cases increase due to the highly contagious Delta variant, Minnesota health officials have reported more demand for monoclonal antibody treatments.
“We do know that metro facilities have been transferring their referrals ... due to lack of infusion appointment availability as all health systems struggle with staffing issues,” said Alexandra Waterman, medical surge coordinator for the Minnesota Department of Health, according to the Star Tribune. “We do expect continued growth in demand.”
At Essentia Health in Duluth, Minn., demand increased from about 10 treatments per week in mid-July to 93 infusions during one week in mid-August.
However, the treatment shouldn’t be viewed as an alternative to vaccines, Razonable warned. He said high-risk patients should seek the monoclonal antibody treatment as soon as possible if they have COVID-19.